Morphogen-IX Ltd, a biotechnology spin-out from Cambridge Enterprise that is focused on the development of bone morphogenetic proteins (BMPs) for the treatment of pulmonary arterial hypertension (PAH), today announced the nomination of its clinical development candidate and initiation of formal preclinical development. The agent, MGX292, is a protein-engineered variant of BMP9 that has proven highly efficacious and safe in extensive preclinical studies.
“We are delighted to announce MGX292 as our drug candidate. It is the result of 3 long years of preclinical research and development by our outstanding team. This drug has real potential to become a new, disease-modifying therapy for patients suffering from pulmonary arterial hypertension. We look forward to the next stage of development leading to clinical trials by 2021.”
Nick Morrell, CEO, Morphogen-IX
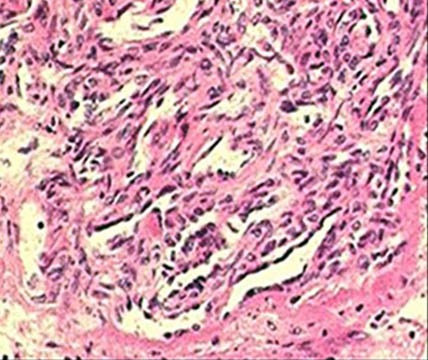
PAH is a rare but devastating disease in which the blood vessels in the lung narrow and close, raising the blood pressure in the lungs, leading to heart failure and death. In many patients the disease has a genetic cause that reduces the protective function of the native BMP9 protein in the blood. MGX292 is the first agent to enter development that can safely restore that protective function for the blood vessels in the lungs.
“The combination of powerful human genetic approaches to identify the importance of BMP9 in PAH and structure-driven engineering of the protein to create a form that can be safely administered has put Morphogen-IX in a world-leading position to develop the first agent capable of halting, or even reversing, the progress of this terrible disease. We are working with many of world’s leading experts in PAH to get our proprietary MGX292 into the clinic as quickly as possible”
David Grainger, Chairman of the Morphogen-IX Board and Chief Scientific Advisor at Medicxi
The full release is available here.